Only A 10th Grade Student Can Pass This Chemistry Quiz
-
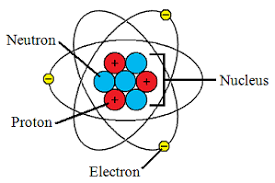
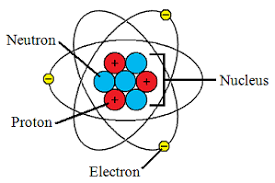
How many electrons are in the first shell of a carbon atom?
-
1
-
2
-
4
-
6
-
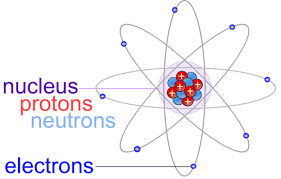
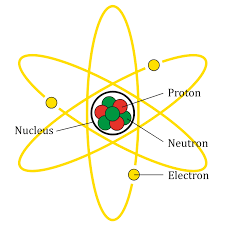
One of the things that Dalton believed was that the atoms of an element were identical to each other. Something evident to you, but was revolutionary at the time. Now we understand why this is the case and can include the number of protons in the definition of an element as follows - atoms that have the same amount of protons are the same element. Can you ace this Atomic Structure quiz?

Quiz Preview
- 2.
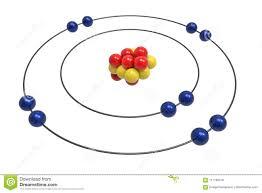
Which element has 2 electrons, 2 neutrons and 2 protons?
-
Fluorine
-
Sodium
-
Helium
-
Neon
Correct Answer
A. HeliumExplanation
Helium is the correct answer because it has 2 electrons, 2 neutrons, and 2 protons. It is the second lightest element in the periodic table and belongs to the noble gas group. Helium is a colorless and odorless gas that is commonly used in balloons and as a coolant in various applications. Its atomic number is 2, indicating that it has 2 protons in its nucleus, which determines its identity as an element. The 2 electrons and 2 neutrons complete the atomic structure of helium.Rate this question:
-
- 3.
An atom of which element has 11 electrons?
-
Potassium
-
Sodium
-
Magnesium
-
Neon
Correct Answer
A. SodiumExplanation
Sodium has 11 electrons because it is located in group 1 of the periodic table, which means it has 1 valence electron. In addition to the valence electron, sodium also has 2 electrons in the first energy level and 8 electrons in the second energy level, totaling 11 electrons. Potassium, magnesium, and neon do not have 11 electrons, so they are not the correct answer.Rate this question:
-
- 4.
How many electrons are in the first shell of a hydrogen atom?
-
1
-
2
-
3
-
4
Correct Answer
A. 1Explanation
The first shell of a hydrogen atom can hold a maximum of 2 electrons. However, since hydrogen only has 1 electron, there is only 1 electron present in the first shell.Rate this question:
-
- 5.
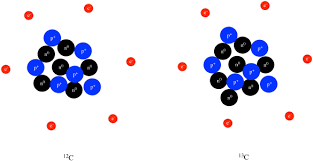
A fluorine atom has 9 electrons - 7 in its outer shell and 2 in its inner shell. How many electrons would be in the outer shell if it were to become a fluoride ion?
-
2
-
4
-
6
-
8
Correct Answer
A. 8Explanation
When a fluorine atom gains an electron to become a fluoride ion, it achieves a stable electron configuration similar to that of a noble gas, in this case, neon. The outer shell of neon contains 8 electrons, so when fluorine gains an electron, it will have 8 electrons in its outer shell, making the correct answer 8.Rate this question:
-
- 6.
An atom has 2 electrons, 2 neutrons and 2 protons. What is the atomic number of this atom?
-
6
-
1
-
2
-
4
Correct Answer
A. 2Explanation
The atomic number of an atom is equal to the number of protons in its nucleus. In this case, the atom has 2 protons, so the atomic number is 2.Rate this question:
-
- 7.
An atom has 7 electrons - 2 in its inner shell and 5 in its outer shell. What group of the periodic table is this element found in?
-
2
-
3
-
4
-
5
Correct Answer
A. 5Explanation
The element with 7 electrons - 2 in the inner shell and 5 in the outer shell belongs to group 5 of the periodic table. Group 5 elements have 5 electrons in their outermost shell and exhibit similar chemical properties due to the same number of valence electrons.Rate this question:
-
- 8.
A fluorine atom has 9 electrons - 7 in its outer shell and 2 in its inner shell. How many electrons would be in the outer shell if it were to become a fluoride ion?
-
2
-
4
-
6
-
8
Correct Answer
A. 8Explanation
When a fluorine atom becomes a fluoride ion, it gains one electron. This electron fills up the outer shell, which can hold a maximum of 8 electrons. Therefore, the fluoride ion would have 8 electrons in its outer shell.Rate this question:
-
- 9.
An element has 2 electrons in its inner shell, 8 in its next shell and 1 in its outer shell. What type of ion would it form?
-
1-
-
1+
-
2+
-
3-
Correct Answer
A. 1+Explanation
The element has 2 electrons in its inner shell, 8 in its next shell, and 1 in its outer shell. Since the outer shell is not complete, it will tend to lose 1 electron to achieve stability. By losing 1 electron, the element will have a net positive charge of 1+, forming a cation. Therefore, the correct answer is 1+.Rate this question:
-
- 10.
An atom contains 9 electrons. How many neutrons are there in the nucleus of this atom?
-
10
-
9
-
19
-
11
Correct Answer
A. 10Explanation
An atom contains a nucleus at its center, which is made up of protons and neutrons. The number of protons determines the atomic number of an element, while the sum of protons and neutrons gives the atomic mass. In this case, the atom contains 9 electrons, so the atomic number is 9. The atomic mass is not given, so we cannot determine the exact number of neutrons. However, since the atomic mass is likely to be close to 19, the closest option is 10 neutrons.Rate this question:
-
- 11.
How many electrons are in the outer shell of a nitride 3- ion?
-
5
-
3
-
8
-
7
Correct Answer
A. 8Explanation
The nitride ion has a charge of 3-, which means it has gained three electrons. Nitrogen, the element in the nitride ion, has five electrons in its outer shell. When it gains three electrons, it fills up its outer shell and now has a total of eight electrons in its outer shell. Therefore, the correct answer is 8.Rate this question:
-
Quiz Review Timeline (Updated): Oct 24, 2024 +
Our quizzes are rigorously reviewed, monitored and continuously updated by our expert board to maintain accuracy, relevance, and timeliness.
-
Current Version
-
Oct 24, 2024Quiz Edited by
ProProfs Editorial Team -
Apr 27, 2019Quiz Created by
Max
Atom Structure And Isotopes: Quiz!
Explore the fundamentals of atomic structure and isotopes with this engaging quiz! Test your knowledge on the relationship between protons, electrons, and neutrons, understand...
Questions:
24 |
Attempts:
2276 |
Last updated:
Sep 02, 2024
|
Atoms And Elements Test Quiz!
The 'Atoms and Elements Test Quiz!' assesses understanding of basic atomic structure, including the role of protons, neutrons, and electrons. It covers atomic number significance,...
Questions:
21 |
Attempts:
1417 |
Last updated:
Oct 30, 2024
|
Chemistry Of The Atom (for Biology Class)
This quiz, titled 'Chemistry of the atom,' assesses key concepts related to atomic structure, focusing on the locations and charges of protons, neutrons, and electrons. It is...
Questions:
8 |
Attempts:
101 |
Last updated:
Feb 23, 2023
|
ATOMIC STRUCTURE - NEET/JEE /KEAM
Each correct answer have 4 Marks
Each wrong answers have -1 Mark
Time - 15 Minutes ( 15 Questions )
Questions:
15 |
Attempts:
261 |
Last updated:
Jul 10, 2024
|
Physics Q1 BIM3 8A
This Physics quiz for BIM3 8A covers fundamental concepts of matter, including atomic structure, physical properties, chemical changes, and states of matter transitions.
Questions:
15 |
Attempts:
101 |
Last updated:
Jan 23, 2023
|
Chemistry Basics: Introduction To Matter Quiz
Explore the building blocks of the universe with our Introduction to Matter Quiz! This comprehensive quiz will challenge your understanding of the basics of chemistry, from...
Questions:
13 |
Attempts:
110 |
Last updated:
May 10, 2024
|
 Back to top
Back to top