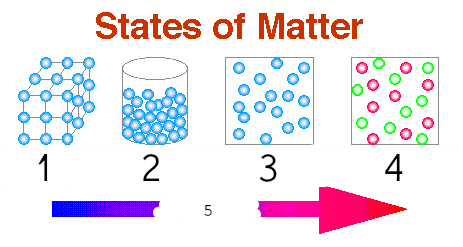
Chemistry Chapter 13: States Of Matter Review
Submit
2.
What first name or nickname would you like us to use?
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
Submit
×
Thank you for your feedback!