Related Flashcards
Cards In This Set
| Front | Back |
|
Name 5 key functions and roles of proteins.
|
- Structure
- Membrane components - Enzymes - Hormones - Antibodies |
|
What are the monomers of proteins?
|
Amino Acids
|
|
How many types of amino acid are there?
|
20
|
|
What determines what amino acid each is?
|
The 'R' Group.
|
|
What is the structure of an amino acid?
|
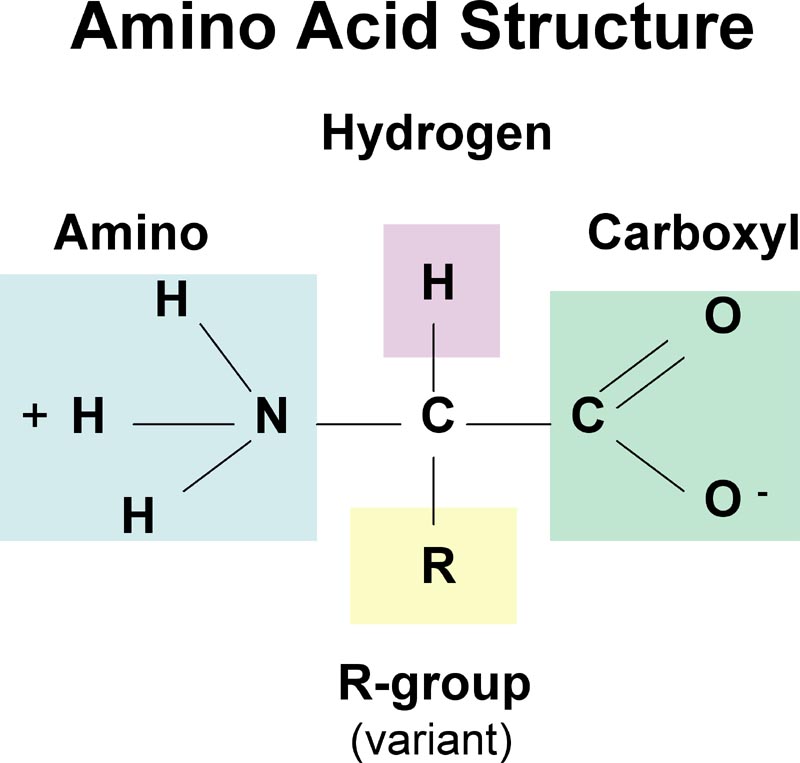 An amino group, an R group, an Acid group and a carbon inbetween. |
|
How do plants and animals obtain amino acids?
|
Plants manufacture them. Animals have to obtain them from thier diet.
|
|
What is deamination, where is it carried out and why?
|
The process of removing the amino group from the amino acid.
This takes place in the Liver and is because excess amino acids cannot be stored by animals as the amino group makes them too toxic if too much is present. |
|
What are amino acids joined by?
|
Condensation.
|
|
What are amino acids split by?
|
Hydrolysis.
|
|
What bond is made?
|
Peptide.
|
|
What is the name of the molecule formed from 2 amino acids?
|
Dipeptide.
|
|
What is the name of the molecule formed from 3 or more amino acids?
|
Polypeptide.
|
|
Why are some amino acids decribed as 'non-essential'?
|
Because they are not essential parts of the diet. They are still essential for protein synthesis but they can be made in the body from other amino acids.
|
|
What is the structure of a dipeptide molecule?
|
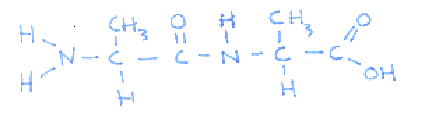 . |
|
Where are polypeptides and proteins synthesised?
|
Ribosomes.
|