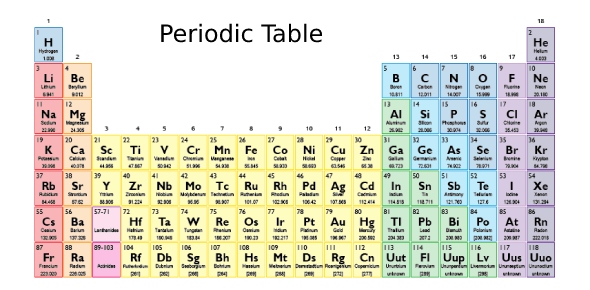
The answer to the question about “Which three groups of the Periodic Table contain the most elements classified as metalloids (semimetals)?” is of 14, 15, and 16. The list of metalloids are as follows: Arsenic, Tellurium, Germanium, Silicon, Antimony, Boron, Polonium, Astatine, Selenium, Aluminium, Carbon, Bismuth, Phosphorus, Beryllium, Tin, Sulfur, Livermorium, Iodine, Flerovium, Gallium, Hydrogen, Lead, Moscovium, and Tennessine.
From the metalloids, group 14 has five metalloids – Carbon, Silicon, Germanium, Tin, and Lead, group 15 has four metalloids – Phosphorus, Arsenic, Antimony, and Bismuth, and group 16 has four metalloids – Sulfur, Selenium, Tellurium, and Polonium. Although groups 13 and 17 also have metalloids, the former only has Boron, Aluminium, and Gallium, while the latter only has Iodine and Astatine.